 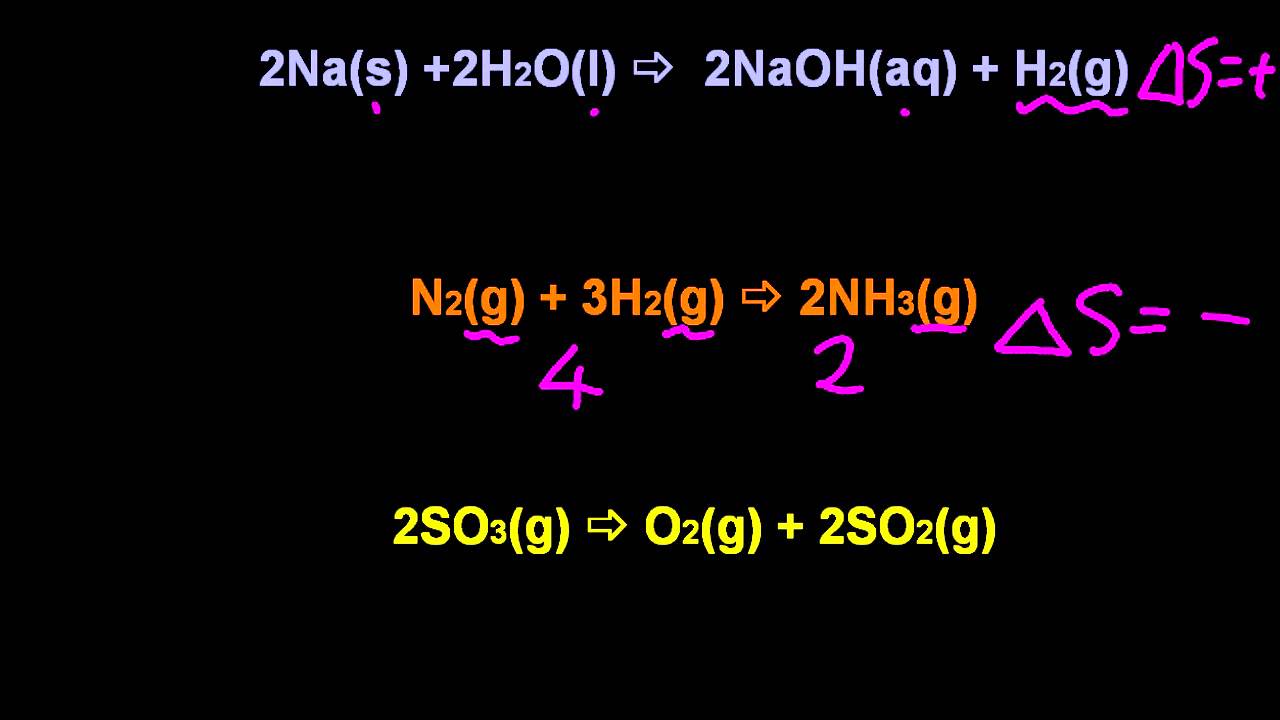
Thus, looking at the activation parameters can reveal a lot about what is going on in the transition state. Dissociative pathway: one molecule converts to two molecules in rate determining step, greater degrees of freedom in two independently moving molecules, entropy increases.Dissociative pathway: more bond breaking in rate determining step, higher enthalpy needs.these two molecules have more degrees of freedom in which to partition their energy than they did as one molecule. Since the entropy of both the system and the surroundings increases, the entropy of the universe must also increase, making the reaction spontaneous at all temperatures. The change n entropy for the reaction is positive the entropy of the system increases (e.g. However, the molecule breaks into two molecules in the rate determining step. The change in enthalpy is negative: heat is emitted. The higher enthalpy of activation results because a bond must be broken in the rate determining step. On the other hand, the dissociative pathway is marked by a higher enthalpy of activation but a positive entropy of activation. Associative pathway: two molecules must be aligned and come together fewer degrees of freedom for energy distribution decrease in entropy.Associative pathway: more bond making than bond breaking lower enthalpy needs.They can no longer move independently of each other, and all of their combined energy must be reapportioned together, with a more limited range of vibrational, rotational and translational states to use for that purpose. The reason the energy must be redistributed this way is that two molecules (the metal complex and the new ligand) are coming together to make one bigger molecule. A positive value indicates an increase in entropy, while a negative value denotes a decrease in the entropy of a system. Usual units of standard molar entropy are joules per mole Kelvin (J/molK). That's not favourable and makes the reaction harder. Standard molar entropy is defined as the entropy or degree of randomness of one mole of a sample under standard state conditions. However, a decrease in entropy means that energy must be partitioned into fewer states. That's favourable and makes the reaction easier. The low enthalpy of activation results because bonds don't need to be broken before the transition state, so it doesn't cost much to get there. These two parameters can be useful in understanding events leading to the transition state.įor example, in ligand substitution, an associative pathway is marked by low enthalpy of activation but a negative entropy of activation. Since is a natural number (1,2,3.), entropy is either zero or positive (ln 1 0, ln 0). The activation entropy deals with how the energy within the molecule must be redistributed for the reaction to occur. The entropy S is proportional to the natural logarithm of this number: The proportionality constant k B is one of the fundamental constants of physics, and is named the Boltzmann constant in honor of its discoverer. The activation enthalpy is the energy required for the reaction. 
In which Δ H ‡ = activation enthalpy and Δ S ‡ = activation entropy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
